<< Back to MOTIFvations Blog Home Page
DNA Methylation’s Impact on cfDNA Fragmentation – Considerations When Choosing Biomarkers
August 29, 2023
Table of Contents:
-
Introduction: The Translational Success of Cell-free DNA
-
Setting the Scene: Nucleosome Accessibility and DNA Methylation Impact Cell-free DNA
-
Getting Down to the Data: DNA methylation Regulates Cell-free DNA Fragmentation
-
Conclusions: DNA Methylation, Cell-free DNA Fragmentation, and Disease Biomarkers in Liquid Biopsies
Introduction: The Translational Success of Cell-free DNA
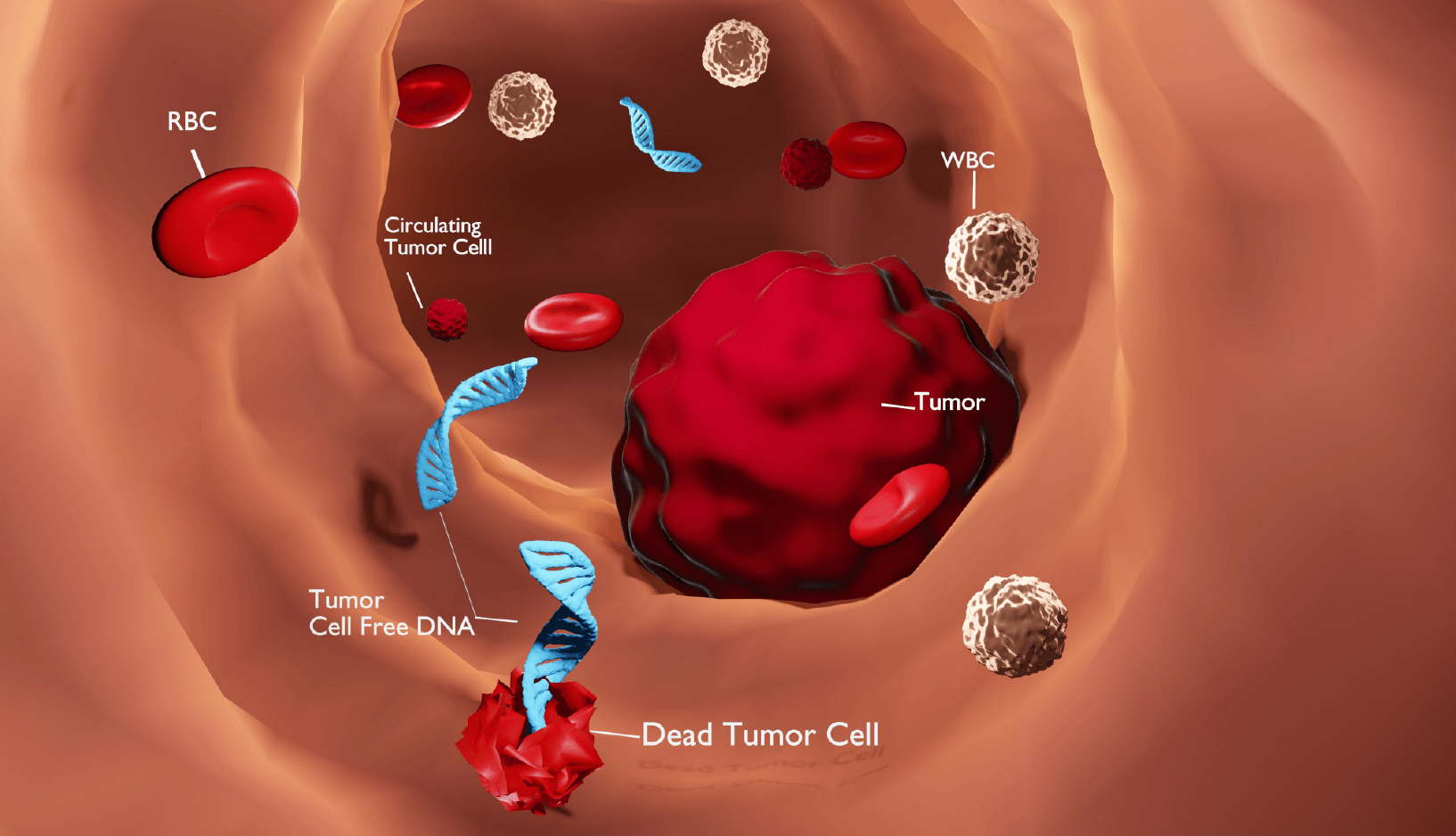
The cell-free DNA molecules present in biological fluids represent an essential component of liquid biopsies and have been employed in non-invasive cancer diagnosis (Stroun et al. and Wan et al.), prenatal testing (Lo et al., 1997), and transplantation monitoring (Lo et al., 1998) to name but a few applications. Cell-free DNA generally exists as short fragments generated through a non-random process (Lo et al., 2021) involving various crucial DNA nucleases (Han et al., 2020, Han et al., 2021 and Han and Lo, 2021).
However, even though the translational power of cell-free DNA has been proven widely, we still understand relatively little regarding the molecular biology of cell-free DNA (Wan et al.), including the underlying principles of cell-free DNA generation and its non-random fragmentation. This knowledge may prove helpful when identifying biomarkers in liquid biopsies for various diseases/conditions.
Setting the Scene: Nucleosome Accessibility and DNA Methylation Impact Cell-free DNA
A previous study (Sun et al.) by researchers from the laboratories of Xin Jin (Beijing Genomics Institute/South China University of Technology) and Kun Sun (Institute of Cancer Research, Shenzhen Bay Laboratory) analyzed the size distribution of Tn5 transposase digested DNA. This research demonstrated that nucleosome accessibility affected cell-free DNA fragment-end cutting preference and that different sizes of cell-free DNA molecules from the plasma of pregnant women become preferentially cut at different positions relative to the nucleosome. In another related study (Lun et al.), the same team of researchers reported some correlation between cell-free DNA size and DNA methylation density.
Now, in their most recent study (An et al.) continue their research trajectory by exploring the underlying molecular bases of cell-free DNA fragmentation by integrating multidimensional functional genomics data. The findings of this exciting cell-free DNA fragmentation study now highlight DNA methylation as a regulator of nuclease cutting preference, which may have broader applications for the identification of biomarkers in cancer liquid biopsies.
Getting Down to the Data: DNA methylation Regulates Cell-free DNA Fragmentation
The authors employed an orientation-aware analysis of cell-free DNA fragmentation patterns against nucleosome structure and integrated multidimensional functional genomics data for healthy controls and colorectal carcinoma patient-derived xenograft mouse models. Their initial findings revealed that cell-free DNA fragment ends for short-sized cell-free DNA molecules displayed drastic differences from those with larger sizes, which suggested the existence of an inherent link between nuclease-mediated end-cutting and cell-free DNA size.
The team also identified a DNA methylation - nuclease preference - cutting end - size distribution axis, which (in short) demonstrated the role of DNA methylation as a functional molecular regulator of cell-free DNA fragmentation. Briefly, cell-free DNA molecules of different DNA methylation levels showed drastically varied sizes and end distributions, with low-level DNA methylation increasing nucleosome accessibility and altering the cutting activities of nucleases during DNA fragmentation, which prompts variation in cutting sites and size distribution of cell-free DNA. Additionally, end motif analysis combined with the findings from other related studies (Han et al., 2020 and Serpas et al.) suggested that the DNASE1L3 (DNASE1L3 deoxyribonuclease 1 like 3) nuclease preferably cuts methylated cell-free DNA during fragmentation.
Finally, and perhaps most interestingly, the authors developed and validated a cell-free DNA fragmentation end-based metric as a promising universal biomarker for pan-cancer diagnosis, demonstrating the potential translational value of this research to cancer liquid biopsy analysis. Overall, this approach may prove helpful for cancer management after large-scale validation studies in the future and the evaluation of the identified metric in combination with additional biomarkers.
Conclusions: DNA Methylation, Cell-free DNA Fragmentation, and Disease Biomarkers in Liquid Biopsies
Overall, these fascinating findings provide a greater understanding of the molecular basis of cell-free DNA fragmentation – through an analysis of the impact of differing DNA methylation levels – to help the definition and study of more powerful biomarkers in cancer liquid biopsies.
For more on how the regulation of cell-free DNA fragmentation by DNA methylation may improve the powers of liquid biopsies, see Nature Communications, January 2023.
About the author

Stuart P. Atkinson, Ph.D.
Stuart was born and grew up in the idyllic town of Lanark (Scotland). He later studied biochemistry at the University of Strathclyde in Glasgow (Scotland) before gaining his Ph.D. in medical oncology; his thesis described the epigenetic regulation of the telomerase gene promoters in cancer cells. Following Post-doctoral stays in Newcastle (England) and Valencia (Spain) where his varied research aims included the exploration of epigenetics in embryonic and induced pluripotent stem cells, Stuart moved into project management and scientific writing/editing where his current interests include polymer chemistry, cancer research, regenerative medicine, and epigenetics. While not glued to his laptop, Stuart enjoys exploring the Spanish mountains and coastlines (and everywhere in between) and the food and drink that it provides!
Contact Stuart on Twitter with any questions
Related Articles
Single-Cell DNA Methylation Sequencing: Small Scale Differences Explain Big Effects!
October 13, 2022
As an epigenetic mark, DNA methylation reveals valuable information on what’s going on inside a cell. In traditional bulk sequencing methods, DNA methylation signals are averaged out so that cell to cell variability in methylation is lost. Single-cell methods for DNA methylation can reveal these hidden signatures. But gathering DNA methylation data on a single-cell level has been challenging. Here we compare methods for scDNA Methylation assays that can uncover valuable insights into cell development, disease, and brain cell heterogeneity. As these have only been around for a short number of years, it’s almost certain that we’ve only just scratched the surface.
Read More
DNA Modifications Reveal the Splendor of the Plant Epigenetic Landscape
July 22, 2022
Would you like to explore the color and beauty of the epigenetic landscape of plants? Begin your journey with a discussion of recent publications highlighting the ongoing discovery and importance of DNA modifications in various plant species.
Read More
<< Back to MOTIFvations Blog Home Page