Chromeo™ 494 is a unique red fluorescent label that is excitable at a wavelength of 488 nm by Argon Laser, LED, Xenon-Arc or Tungsten-Halogen lamps. It has a very large Stokes shift of 134 nm, making it an ideal partner for multiplexing in immunofluorescence experiments, especially in combination with Chromeo 488. While both dyes are readily excited at 488 nm, the fluorescent emission of Chromeo 488 at 517 nm is easily distinguished from the emission of Chromeo 494 at 628 nm. This long Stokes shift eliminates signal overlap caused by bleed-through between the detection channels in multiplexing experiments.
Why use Chromeo™ 494?
- Large Stokes shift – ideal for multiplexing with other 488-excitable dyes
- Photostability
- Low background
- pH insensitive
- Easy to use – no harsh chemicals required for conjugation
Reactive fluorescent Chromeo 494 serves as a bright label for antibodies and other biomolecules, enabling detection in immunofluorescence, high content screening, ELISA and FRET applications, as well as in flow cytometry.
To ensure the highest quality in your immunofluorescent staining, we recommend the use of Chromeo 494 together with our complete line of MAX Stain™ Immunofluorescence Tools, as those components have been formulated to optimize the performance of all Fluorescent Chromeo™ Dyes. In addition we recommend the use Mowiol or TDE (2,2'-thiodiethanol) to mount the slide preparations, as these media have been shown to increase the quality of fluorescence microscopy images using Chromeo 494.
| Name | Format | Cat No. | 価格 (税抜) | |
|---|---|---|---|---|
| Chromeo™ 494 Alkyne | 1 unit | 15190 | Get Quote | |
| Chromeo™ 494 Azide | 1 unit | 15193 | Get Quote | |
| Chromeo™ 494 NHS-Ester | 1 unit | 15196 | Get Quote |
| Chromeo™ Fluorescent Dyes, Secondaries & Assays Profile |
| Cell Biology Products Brochure |
| Chromeo™ 494 Alkyne Data Sheet |
| Chromeo™ 494 Azide Data Sheet |
| Chromeo™ 494 NHS-Ester Data Sheet |
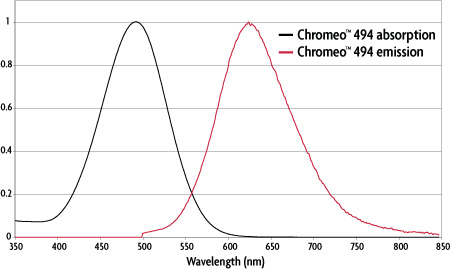
Figure 1: Absorption and emission spectra of Chromeo™ 494.
| Dye | Absorption | Emission | ε L/(mol-cm) | Stokes shift | |
|---|---|---|---|---|---|
| Chromeo™ 494 | 494 nm | 628 nm | 55,000 | 134 nm | |
Table 1: Properties of Chromeo™ 494. |
|||||
Chromeo™ 494 is a unique fluorescent dye that can be excited at 488 nm. Due to its large Stokes shift of 134 nm, the dye emits a red fluorescence. Chromeo 494 is compatible with all excitation sources used for fluorescein, FITC, Chromeo 488 and other 488 nm-excitable dyes. However, because the red emission of Chromeo 494 is separated completely from the green fluorescence emitted by other 488-excitable dyes, Chromeo 494 and Chromeo 494 dyes are ideal partners for green fluorescent dyes and green fluorescent proteins in multi-color experiments.
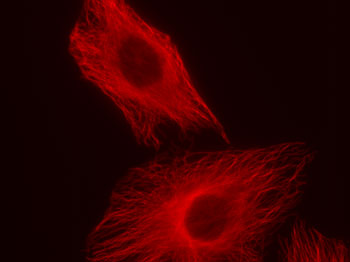
Figure 1: Chromeo 494 staining in HeLa cells.
HeLa cells were stained with rabbit alpha Tubulin polyclonal antibody and Chromeo 494 Goat anti-rabbit IgG.
The fluorescent properties of Chromeo 494, especially its long Stokes shift, and its ability to be combined with MAX Stain™ Immunofluorescence Tools make Chromeo 494 and its Fluorescent Secondary Antibody Conjugates excellent partners of Chromeo 488 and other 488 nm-excitable dyes for use in fluorescence microscopy and multiplexing experiments.
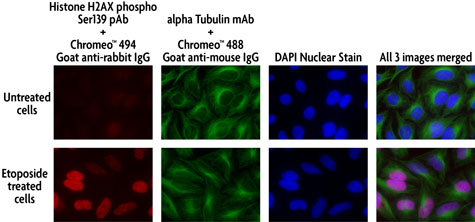
Figure 2: Multi-color staining of Phospho H2AX and tubulin using Chromeo 494 and Chromeo 488 secondary antibodies in untreated and ETO-treated HeLa cells.
HeLa cells were either left untreated or treated with 100 µM ETO for 6 hours prior to fixation with methanol. The histone-variant H2AX was stained with Histone H2AX phospho Ser139 rabbit pAb (Catalog No. 39117) and Chromeo 494 Goat anti-rabbit IgG, while tubulin was visualized using alpha Tubulin mouse mAb (Clone 5-B-1-2) and Chromeo 488 Goat anti-mouse IgG.
Chromeo 494 has been shown to work in several different types of fluorescence microscopy. In addition to classical widefield applications, Chromeo 494 and its Fluorescent Secondary Antibody Conjugates have been tested in confocal scanning microscopy and high resolution microscopy. The fluorescent properties of Chromeo 494 dye and secondaries meet the specifications required to perform dual-color STED microscopy, as demonstarted by co-localization experiments in the cell nucleus (Figure 3), in synaptic complexes of dendritic cells (Figure 4) or by staining tubulin (Figure 5).
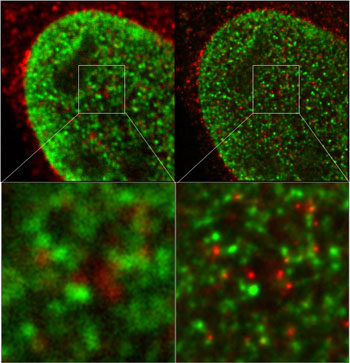
Figure 3: Nuclear structures visualized by dual color STED experiments.
The image on the left was prepared using a confocal microscope, while that on the right was produced using a STED microscope. The nuclear structures have been visualized with Chromeo 494 (green) and ATTO 647N (red). Images courtesy of Dr. L. Schermelleh, LMU Biozentrum Munich, Germany.
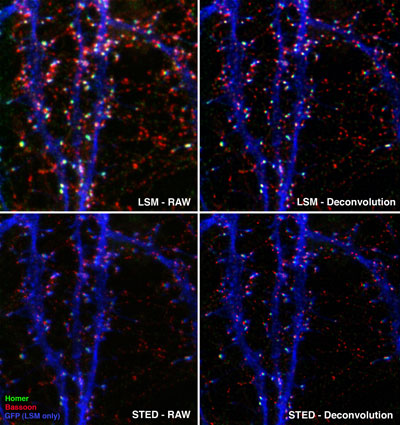
Figure 4: Pre- and post-synaptic marker proteins visualized by STED microscopy.
The localization of pre- and post-synaptic marker proteins visualized in the dendrites of nerve cells. The pre-synaptic protein Bassoon was stained by ATTO 647N (red), while the post-synaptic protein Homer was stained with Chromeo 494 (green). The dendrite structure has been visualized by GFP and is presented as a blue overlay of a classical confocal picture. This STED image is courtesy of Dr. W. Zuschratter, IfN Magdeburg, Germany.
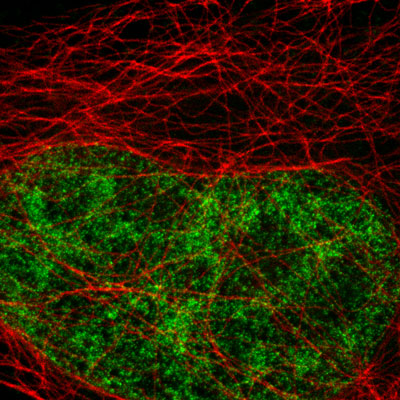
Figure 5: Active Motif's primary antibodies and fluorescent secondary antibodies in STED microscopy.
HeLa cells were stained with alpha Tubulin mouse monoclonal antibody (Clone 5-B-1-2) and Chromeo 494 Goat anti-mouse IgG. Histone H3 was stained with Histone H3 trimethyl Lys4 rabbit polyclonal antibody (Catalog No. 39159) and the ATTO 647N Goat anti-rabbit IgG secondary antibody. The STED image is courtesy of Leica Microsystems, Germany.
The following selected publications cite the use of and/or provide additional information about Chromeo 494:
- “Development of an RNA interference method in the cladoceran crustacean Daphnia magna” by Kato et al (2011) Dev Genes Evolution Nos. 11-12:337-345.
- “Two-color STED microscopy in living cells” by Pellett et al (2011) Biomed. Optics Express 2(8):2364-2371.
- “Primitive Endoderm Differentiates via a Three-Step Mechanism Involving Nanog and RTK Signaling” by Frankenberg et al (2011) Developmental Cell 21:1005-1013.
- “Fast neurotransmitter release regulated by the endocytic scaffold intersectin” by Sakaba et al (2013) PNAS 110(20):8266-8273.
- “A mutation in the receptor Methoprene-tolerant alters juvenile hormone response in insects and crustaceans” by Miyakawa et al (2013) Nature Comm. 4: Artice number: 1856.
- “Super-resolution Microscopy of the Neuronal Calcium-Binding Proteins Calneuron-1 and Calendrin” by Hradsky et al (2013) Methods in Mol Biol. 963: 147-167.

